Why is oxygen 8th on the periodic table
 Robert Guerrero
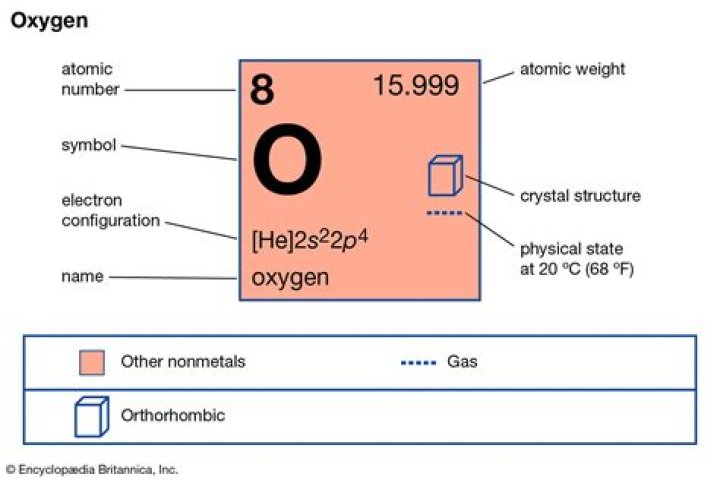
Robert Guerrero On the periodic table of elements, oxygen is represented by the symbol O. It has an atomic number of 8 because it has 8 protons in its nucleus.
Why is oxygen the 8th element on the periodic table?
Oxygen with the symbol O has the atomic number 8 which means it is the 8th element in the table. The number eight also means that oxygen has eight protons in the nucleus. The number of protons and the number of electrons are always the same in an element that is neutral and has no charge.
What is the 8th element on the periodic table?
Oxygen – Element information, properties and uses | Periodic Table.
Is oxygen 8 on the periodic table?
Hooray for oxygen, the element that keeps much of life on Earth humming. Element No. 8 on the Periodic Table of the Elements is a colorless gas that makes up 21 percent of Earth’s atmosphere.Does oxygen have 8 electrons?
An oxygen atom usually has 8 protons, 8 neutrons, and 8 electrons. Looking at the periodic table, oxygen has atomic number 8 and atomic weight 15.999.
Where is oxygen on the periodic table?
oxygen (O), nonmetallic chemical element of Group 16 (VIa, or the oxygen group) of the periodic table.
What atom has 8 protons 8 electrons and 9 Neutrons?
8 17 This isotope of oxygen has 8 protons (atomic number), 8 electrons and 9 neutrons. (Recall that mass number is the total of protons and neutrons. ) Isotopes are also often written as C-12 C-13 C-14 The number is the mass number.
What element contains 8 protons in its nucleus?
Oxygen by definition has 8 protons in its nucleus.What element has 8 protons 8 neutrons and 10 electrons?
16O2− has 8 protons, 8 neutrons, and 10 electrons.
Why can't oxygen have 9 protons?An oxygen atom has 8 protons in its nucleus. Why can’t it have 9 protons? Because an atom that has 9 protons would be Fluorine.
Article first time published onHow many elements make up oxygen o2?
Under standard conditions oxygen forms a gas that is composed of molecules consisting of two oxygen atoms (O2). This is called a diatomic gas. In this form oxygen is a colorless, odorless, tasteless gas. Oxygen also exists as the allotrope ozone (O3).
What are the 8 periodic table groups?
- Alkali metals.
- Alkaline earth metals.
- Rare earth metals.
- Crystallogens.
- Pnictogens.
- Chalcogens.
- Halogens.
- Noble gases.
What are Group 8 elements used for?
The noble gases are used to form inert atmospheres, typically for arc welding, to protect specimens, and to deter chemical reactions. The elements are used in lamps, such as neon lights and krypton headlamps, and in lasers. Helium is used in balloons, for deep-sea diving air tanks, and to cool superconducting magnets.
Can oxygen have any other atomic number than 8?
For example, there are three isotopes of the element oxygen (O): Oxygen 16, 17, and 18. Each isotope of oxygen contains 8 protons, but differs in the number of neutrons.
Which of the following is true of oxygen that has 8 protons 8 neutrons and 8 electrons?
Which of the following is true of oxygen that has 8 protons, 8 neutrons, and 8 electrons? It has an atomic number of 8. Which of the following has the smallest total mass?
What isotope has the atomic number 8?
Oxygen, element symbol O, is the element that is atomic number 8 on the periodic table. This means every atom of oxygen has 8 protons. Varying the number of electrons forms ions, while altering the number of neutrons makes different isotopes of the element, but the number of protons remains constant.
What is the mass number of an oxygen isotope that consists of 8 protons 8 electrons and 9 Neutrons?
oxygen-17 has 8 protons and electrons. # neutrons = mass # – #protons. So, in oxygen-17, # neutrons = 17 – 8 or 9. Oxygen-17 has 8 protons, 8 electrons, and 9 neutrons.
Is oxygen charged?
Organic Chemistry The oxygen atom is slightly negatively charged, and the carbon and hydrogen atoms are slightly positively charged. The polar bonds of the hydroxyl group are responsible for the major reaction characteristics of alcohols and phenols.
Why is oxygen an element?
Oxygen is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is O, and its atomic number is 8, which means that an oxygen atom has eight protons in its nucleus. Two oxygen atoms strongly bind together with a covalent double bond to form dioxygen or O2. …
What is oxygen explain?
Oxygen is a chemical element with an atomic number of 8 (it has eight protons in its nucleus). Oxygen forms a chemical compound (O2) of two atoms which is a colorless gas at normal temperatures and pressures. … About 21% of Earth’s atmosphere is oxygen.
Where does oxygen come from?
At least half of Earth’s oxygen comes from the ocean. Scientists estimate that 50-80% of the oxygen production on Earth comes from the ocean. The majority of this production is from oceanic plankton — drifting plants, algae, and some bacteria that can photosynthesize.
What is the neutrons of oxygen?
An oxygen atom has 8 positive protons and 8 neutrons in its nucleus, and 8 negative electrons speeding around the nucleus.
How many protons does oxygen have in the nucleus?
The number of protons an atom has is called its Atomic Number. The atomic number of carbon is 6 because carbon atoms have 6 protons. The atomic number of oxygen is 8 because oxygen atoms have 8 protons in their nucleus. The atomic number of gold is 79 because gold atoms have 79 protons.
What is the nucleus of oxygen atom?
Oxygen is atomic number 8. Therefore, its nucleus contains 8 protons. Protons are positively charged. One proton has a charge of +1 .
How many electrons does oxygen have in its outer shell?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell. We can write the configuration of oxygen’s valence electrons as 2s²2p⁴.
What element has 6 protons 8 neutrons?
For example, carbon-14 is a radioactive isotope of carbon that has six protons and eight neutrons in its nucleus. We call it carbon-14 because the total number of protons and neutrons in the nucleus, also known as the mass number, adds up to 14 (6+8=14).
What element contains 7 protons?
If it has 7 protons, that’s nitrogen. But the logic of the periodic table also relies upon the number and organisation of electrons in an atom. Let’s take chlorine as an example. It is positioned in group 7 of the periodic table because it has seven electrons in the outermost shell of electrons.
What element has 10 protons 10 electrons and 10 neutrons?
Neon is an atom with atomic number ten. Its atomic weight is 20.179 which cause it to have ten neutrons and ten protons in its nucleus and ten electrons outside.
Why is oxygen O2 and not just O?
Oxygen is available as O2 not as O because pure oxygen is highly reactive it will combine with anything to form oxides,disconnected atoms have a very strong tendency to form bonds. So if there is nothing else available for oxygen atom to combine it will combine with another oxygen atom to form an oxygen molecule.
What is oxygen made out of?
Oxygen is a non-metal element and is found naturally as a molecule. Each molecule is made up of two oxygen atoms that are strongly joined together. Oxygen has low melting and boiling points, so it is in a gas state at room temperature.
Why oxygen is the most abundant element?
Oxygen is the most abundant element on Earth because oxygen is the third most abundant element in the universe and it is extremely chemically reactive, so it is bound up in all kinds of materials. The most abundant elements in the universe by far are hydrogen and helium, most of which are in stars.